“90% efficacy” means if only they’d had the vaccine, on average only 10 would have got ill. The primary way the effectiveness of a vaccine is assessed is through its “efficacy.” This can be explained by imagining that 100 people are ill with COVID. The level of antibodies created by your body following the vaccine is measured using this test. The two main vaccines available in the UK: Pfizer/BioNTech and AstraZeneca, both aim to create IgG antibodies to the spike protein of this coronavirus. Testing For All will continue to publish data on the immune response created by the different vaccines available in the UK. 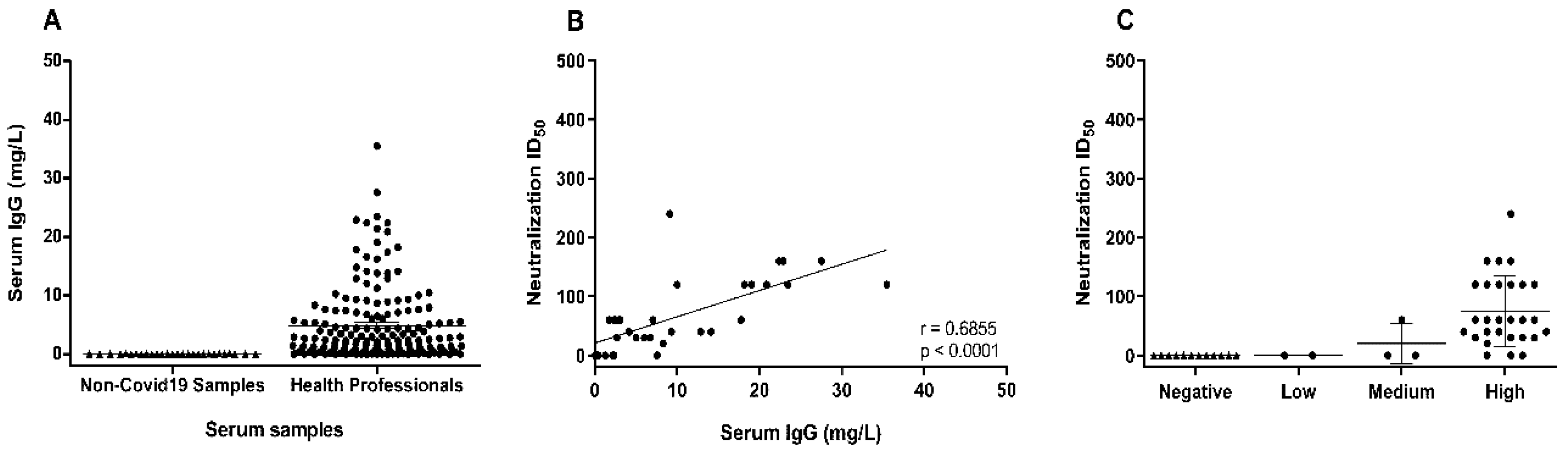
Research indicates that vaccination can fail due to either the way the vaccine was manufactured and handled, or due to the host’s immune response. One of the primary use cases for this test is to check that your immune system has responded correctly to the vaccine and produced antibodies in the nominal range. The sections below will be updated shortly to include the break down by number of doses, and immune status. In the period 12th Feb to 12th June 2021 Testing For All completed 3,016 quantitative Anti-SARS-CoV-2 (S) tests. Those that receive a result of > 2,500 have a neutralising antibody response of at least 2,500 U/mL indicating a very high antibody level. The Roche Anti-SARS-CoV-2 (S) test will return “ 2,500” or greater than 2,500. The difference between the two tests is that they are looking at different viral protein targets, and that the Immunity Test gives you a calibrated antibody titer that can be compared to other tests and the emerging WHO standard. In the testing that we have carried out with our laboratory partner, we have compared the result of the both the Roche N and S test tests on positive blood samples and see very good agreement so if you tested positive on our COVID-19 Home Total Antibody Test then you will very likely test positive on the COVID-19 Immunity Test. The COVID-19 Immunity Test provides an accurate calibrated measurement of the level of antibodies to the spike protein, and is therefore considered to be a good view of your immune response and status. The original COVID-19 Home Total Antibody Test we launched in Sept 2020 detects antibodies to the Nucleocapsid protein that surrounds the RNA of the coronavirus, and is useful for detecting previous exposure, which has provided many people with an understanding of whether past or ongoing symptoms that they experienced were related to a coronavirus infection that occurred before PCR testing was widely available. A natural infection creates a wide ranged defence, often involving a T cell response to destroy human cells infected by SARS-SoV-2 to stop virus from replicating, and by the creation of a range of antibodies to different proteins in the virus that attempt to deactivate it before it can bind to host cells. These “spikes” are what gives coronaviruses their name, and it is through this spike mechanism that the virus binds to healthy human cells in order to replicate itself. The body has a range of ways to fight off infection, but the scientific community has centred the development of vaccines for COVID on triggering the creation of antibodies to the spike protein of coronavirus. For those that wish to, this test can also be used to monitor your antibody level (or antibody titer as it is more formally known) over time as the test provides a calibrated result in U/mL that will change as your IgG antibody level builds and then wanes over an extended period of time. The aim of this product is to help the Testing For All community to understand their immune response, and hence their immunity status to COVID-19. This article explains the result from the Roche Anti-SARS-CoV-2 (S) test to give Testing For All users a better understanding of how this diagnostic test works.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
